Good links for Unit 1 from SSERC:
https://www.sserc.org.uk/subject-areas/chemistry/cfe-advanced-higher-chemistry/
Electromagnetic Spectrum

Dual nature of electromagnetic radiation -
- Fun video - bit confusing at the end but at least that is physics rather than chemistry at the end! https://www.youtube.com/watch?v=DfPeprQ7oGc
- Lecture - https://www.youtube.com/watch?v=RlPvzn5sG30
- Very interesting photograph which shows waves in light using an electron microscope: http://www.livescience.com/50019-image-light-wave-particle.html
Electromagnetic Radiation - Revision of Basics
An entertaining presentation and revision of the basics of Electromagnetic Radiation (is that Matt Baker speaking ?!?) :
http://www.bbc.co.uk/schools/gcsebitesize/science/edexcel/electromagnetic_spectrum/electromagneticspectrumact.shtml
Dual Nature of Electromagnetic Radiation :Is it like a wave or particle or both ?
Some Nobel Prizes which were based on this are discussed here :
http://www.nobelprize.org/nobel_prizes/themes/physics/ekspong/
Spectroscopy in a Suitcase - some interesting resources, particularly for some background reading - http://www.rsc.org/learn-chemistry/resource/res00000280/spectroscopy-in-a-suitcase-students-resource?cmpid=CMP00000320
Energy levels in an atom
remember the energy levels are spaced more like this with a bigger difference in energy between n=1 and n=2 than for n=2 and n=3 etc etc
The maximum number of electrons possible in the first four energy levels are:
n= 1 2 electrons
n = 2 8 electrons
n = 3 18 electrons
n = 4 32 electrons
There are 4 subshells: s, p, d and f
subshell s can have 2 electrons
subshell p can have 6 electrons
subshell d can have 10 electrons
and subshell f can have 14 electrons
Orbitals and Subshells
Hydrogen Spectrum
Here are the transitions between the various electron energy levels (the arrows!!) which result in the four coloured lines on the visible part of the Atomic Emission spectrum for hydrogen :
Emission and Absorption spectra of Hydrogen Helium Lithium and Beryllium :
http://scisyn.com/umuc/astro/ASTR100Notes/spectra-examples.html
"William Ramsay."
from Famous Scientists. famousscientists.org. 19 Dec. 2016. Web. 6/17/2020
www.famousscientists.org/william-ramsay/
Re: Helium - it was Norman Lockyer who pointed the spectroscope at the sun and saw the lines indicating Helium (named after Helios for sun..) ..he was initially ridiculed but then William Ramsay discovered Helium properly and proved it existed !
And some more about Norman Lockyer:
http://normanlockyer.com/history/spectral-plates/
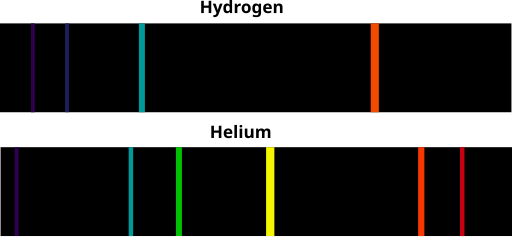
(Image ... Ranjithsiji / CC BY-SA (https://creativecommons.org/licenses/by-sa/4.0)
https://commons.wikimedia.org/wiki/File:Atomic_emission_spectrum_of_helium.svg)
Why you get colour from salts of Alkali metals
RSC Video of Christmas lecture - very good
Using/Making a simple CD Spectroscope
You can make a very simple spectroscope from a CD (or even just hold one up to the light), to analyse emissions from lights such as neon / sodium vapour streetlight, candle light etc. Details of making one are here and examples of what you might see :
https://www.cs.cmu.edu/~zhuxj/astro/html/spectrometer.html
or http://www.bbc.co.uk/schools/gcsebitesize/science/edexcel/waves_universe/exploring_spacerev6.shtml
So many words !! Spectroscope vs Spectrometers vs Spectrophotometer
You may come across all these words but essentially they all allow you to view emission or absorption spectra across all the wavelengths and some allow you to measure and graph the results as well. Different from a colorimeter which you will come across later which measures at one particular wavelength only - although you can use a spectrometer to do that as well!
Wavenumber - you might need to know this too https://www2.chemistry.msu.edu/faculty/harrison/cem483/wavenumbers.pdf
Flame Tests
Group 1 Elements - video of flame tests
http://www.rsc.org/learn-chemistry/resource/res00001120/ri-christmas-lectures-2012-group-1-flame-tests?cmpid=CMP00002100#!cmpid=CMP00002276
In the colours seen, we see energy emitted at a discrete energy level as electrons move from a high energy level to a lower energy level, emitting a discrete (definite) amount of energy which we perceive as a particular colour. Can also be viewed through a spectroscope to see the discrete levels.
How to do a Flame Test -
Using a wooden splint or toothpick? http://www.rsc.org/learn-chemistry/resource/res00000759/flame-tests-the-wooden-splint-method
Another flame test video :
.....and some more !
(I love flame tests !!)
Slightly hesitant explanation but nice to see the different colours from the lamps and flame tests of alkali metals. Only 3 mins long! http://www.rsc.org/learn-chemistry/resource/res00001248/the-alkali-metals-part-1#!cmpid=CMP00002461
Emission and Absorption Spectra :
"What is Spectroscopy"
This page shows a Hydrogen Absorption and Emission Spectrum in Visible light.
http://coolcosmos.ipac.caltech.edu/cosmic_classroom/ir_tutorial/spec.html
If you want to read more and in particular the physics of all this then here is a link to a book you might like - but this is mostly not necessary for Adv Chemistry !
Sodium Lamps - arc lamps
 A sodium street lamp is an arc lamp which is an electric lamp in which a current travels through a gas between two incandescent electrodes and generates an arc that produces light. Also called arc light. The commonly seen Sodium street lamp actually also contains Neon which glows red as the lamp is starting up. Once the lamp has been on for about 10 minutes or so, you will see the orange-yellow glow of the Sodium emission. Find out more from this video about how this lamp works. Interesting to listen to - http://www.edisontechcenter.org/SodiumLamps.html
A sodium street lamp is an arc lamp which is an electric lamp in which a current travels through a gas between two incandescent electrodes and generates an arc that produces light. Also called arc light. The commonly seen Sodium street lamp actually also contains Neon which glows red as the lamp is starting up. Once the lamp has been on for about 10 minutes or so, you will see the orange-yellow glow of the Sodium emission. Find out more from this video about how this lamp works. Interesting to listen to - http://www.edisontechcenter.org/SodiumLamps.html
Lighting - Lots about different types of lamps and lighting- just if you are interested - not really Adv Higher Chemistry except for the sodium Arc lamps perhaps!
http://www.edisontechcenter.org/ArcLamps.html
Atomic Absorption Spectroscopy and Atomic Emission Sepctroscopy can be used quantitatively (ie to measure how much of something there is) and qualitatively (to find out exactly what that thing is)-
http://www.andor.com/learning-academy/atomic-spectroscopy-atomic-absorption,-emission-and-fluorescence-techniques
Working out questions on this ... using the formulae from data booklet:
Orbitals
Nice link about s orbitals, p orbitals etc http://chemguide.co.uk/atoms/properties/atomorbs.html
Quantum Numbers
Each electron in an atom can be defined by its own set of quantum numbers - each electron has a different position ....
n = the principal quantum number = the shell
l = the angular momentum quantum number = the type of orbital eg 0 = s, 1 = p, 2 = d ....
ml = magnetic quantum number = which orbital... eg -1, 0 , 1 for p orbitals...
ms = spin quantum number = which electron in the orbital - 0.5 or + 0.5
etc.
Periodic Table
An interactive periodic table which allows you to select different elements to see if their outer orbital is an s, p or d orbital. Its useful to work through from the beginning and remember that its the outer layers that you are seeing :
http://www.rsc.org/periodic-table/
Also you can use this table to check the spectroscopic notation for each element.
Shapes of Molecules
You can make a very simple spectroscope from a CD (or even just hold one up to the light), to analyse emissions from lights such as neon / sodium vapour streetlight, candle light etc. Details of making one are here and examples of what you might see :
https://www.cs.cmu.edu/~zhuxj/astro/html/spectrometer.html
or http://www.bbc.co.uk/schools/gcsebitesize/science/edexcel/waves_universe/exploring_spacerev6.shtml
So many words !! Spectroscope vs Spectrometers vs Spectrophotometer
You may come across all these words but essentially they all allow you to view emission or absorption spectra across all the wavelengths and some allow you to measure and graph the results as well. Different from a colorimeter which you will come across later which measures at one particular wavelength only - although you can use a spectrometer to do that as well!
- In case you come across Wavenumber here is a wavenumber/wavelength converter http://www.impublications.com/wavenumber-wavelength-converter
Group 1 Elements - video of flame tests
http://www.rsc.org/learn-chemistry/resource/res00001120/ri-christmas-lectures-2012-group-1-flame-tests?cmpid=CMP00002100#!cmpid=CMP00002276
In the colours seen, we see energy emitted at a discrete energy level as electrons move from a high energy level to a lower energy level, emitting a discrete (definite) amount of energy which we perceive as a particular colour. Can also be viewed through a spectroscope to see the discrete levels.
How to do a Flame Test -
Using a wooden splint or toothpick? http://www.rsc.org/learn-chemistry/resource/res00000759/flame-tests-the-wooden-splint-method
Another flame test video :
.....and some more !
(I love flame tests !!)
- https://www.youtube.com/watch?v=9oYF-HxtoYg
- https://www.youtube.com/watch?v=EbkLp3-o78E
- and flame test bottles - your teacher might do this one if you are lucky ! but stand well back! https://www.youtube.com/watch?v=p8imh_yU0Kc
Slightly hesitant explanation but nice to see the different colours from the lamps and flame tests of alkali metals. Only 3 mins long! http://www.rsc.org/learn-chemistry/resource/res00001248/the-alkali-metals-part-1#!cmpid=CMP00002461
Emission and Absorption Spectra :
"What is Spectroscopy"
This page shows a Hydrogen Absorption and Emission Spectrum in Visible light.
http://coolcosmos.ipac.caltech.edu/cosmic_classroom/ir_tutorial/spec.html
If you want to read more and in particular the physics of all this then here is a link to a book you might like - but this is mostly not necessary for Adv Chemistry !
Sodium Lamps - arc lamps
 A sodium street lamp is an arc lamp which is an electric lamp in which a current travels through a gas between two incandescent electrodes and generates an arc that produces light. Also called arc light. The commonly seen Sodium street lamp actually also contains Neon which glows red as the lamp is starting up. Once the lamp has been on for about 10 minutes or so, you will see the orange-yellow glow of the Sodium emission. Find out more from this video about how this lamp works. Interesting to listen to - http://www.edisontechcenter.org/SodiumLamps.html
A sodium street lamp is an arc lamp which is an electric lamp in which a current travels through a gas between two incandescent electrodes and generates an arc that produces light. Also called arc light. The commonly seen Sodium street lamp actually also contains Neon which glows red as the lamp is starting up. Once the lamp has been on for about 10 minutes or so, you will see the orange-yellow glow of the Sodium emission. Find out more from this video about how this lamp works. Interesting to listen to - http://www.edisontechcenter.org/SodiumLamps.htmlLighting - Lots about different types of lamps and lighting- just if you are interested - not really Adv Higher Chemistry except for the sodium Arc lamps perhaps!
http://www.edisontechcenter.org/ArcLamps.html
Atomic Absorption Spectroscopy and Atomic Emission Sepctroscopy can be used quantitatively (ie to measure how much of something there is) and qualitatively (to find out exactly what that thing is)-
http://www.andor.com/learning-academy/atomic-spectroscopy-atomic-absorption,-emission-and-fluorescence-techniques
Working out questions on this ... using the formulae from data booklet:
Orbitals
Nice link about s orbitals, p orbitals etc http://chemguide.co.uk/atoms/properties/atomorbs.html
Quantum Numbers
Each electron in an atom can be defined by its own set of quantum numbers - each electron has a different position ....
n = the principal quantum number = the shell
l = the angular momentum quantum number = the type of orbital eg 0 = s, 1 = p, 2 = d ....
ml = magnetic quantum number = which orbital... eg -1, 0 , 1 for p orbitals...
ms = spin quantum number = which electron in the orbital - 0.5 or + 0.5
etc.
Periodic Table
An interactive periodic table which allows you to select different elements to see if their outer orbital is an s, p or d orbital. Its useful to work through from the beginning and remember that its the outer layers that you are seeing :
http://www.rsc.org/periodic-table/
Also you can use this table to check the spectroscopic notation for each element.
Shapes of Molecules
VSEPR
Explore the shames of the molecules here:
Spin a 3D Methane molecule
https://www.chemtube3d.com/vseprshapech4/
Water:
https://www.chemtube3d.com/vsepr-shape-water-is-bent/
https://www.chemtube3d.com/vseprshapech4/
Water:
https://www.chemtube3d.com/vsepr-shape-water-is-bent/
Shapes of Molecules
Discover which molecules take up certain typical shapes :
http://www.rsc.org/education/teachers/resources/databook/int_shapes_molecules_ions.htm
Colorimetry - Atomic Absorption Spectroscopy
www.docbrown.info/page07/appendixtrans09.htm
If your school doesn't have a colorimeter then here is a method using simple electronics which could easily be bought and your school will surely have some of these parts. All you need to buy are some LED light bulb emitters and receivers and find some LEGO bricks would you believe! https://www.youtube.com/watch?v=RgiIxqBmt1E (from the CLEAPSS website)
This is very clever and simple to try at home if you download a light meter App to your mobile phone - From Royal Society of Chemistry Where he talks of Lux numbers here that is the amount of light that has transmitted through the coloured liquid. The higher the lux number the lower the Absorbance value - which is the value we normally refer to for Advanced Higher …
Some experiments to try using Colorimetry:
Calcium content in milk - complexometric titration and colorimetry: https://www.canterbury.ac.nz/media/documents/science-outreach/calcium.pdf
Beer Lambert Law / Absorbance - good explanation - though may be a little more information than you need for Adv H Chemistry ....Go to Reference
A bit about Colour !
In chemistry and pigments, the following happens ...
http://www.mikeblaber.org/oldwine/chm1045/notes/Bonding/Resonan/Bond07.htm
Interesting film about Forensic Science
Physics solving Crimes ! Using an Ion beam of X-rays and Gamma rays to obtain a spectrum of gunshot residue :
http://www.youtube.com/watch?v=JgbdaQWfjDk
Transition Metal compounds
Colourful manganese and vanadium compounds - colour varies depending on oxidation state of the compounds they are involved in:
Colour changing in Manganese
Vanadium colour changes
The Colours of Complex Metal Ions
This is worth printing out to keep and read:
Coloured Glass - another nice picture to print out : http://www.compoundchem.com/wp-content/uploads/2015/03/The-Chemistry-of-Coloured-Glass.png
- a video about colouring glass - https://www.youtube.com/watch?v=FJ4PGwDtSMs
Discover which molecules take up certain typical shapes :
http://www.rsc.org/education/teachers/resources/databook/int_shapes_molecules_ions.htm
Colorimetry - Atomic Absorption Spectroscopy
www.docbrown.info/page07/appendixtrans09.htm
If your school doesn't have a colorimeter then here is a method using simple electronics which could easily be bought and your school will surely have some of these parts. All you need to buy are some LED light bulb emitters and receivers and find some LEGO bricks would you believe! https://www.youtube.com/watch?v=RgiIxqBmt1E (from the CLEAPSS website)
This is very clever and simple to try at home if you download a light meter App to your mobile phone - From Royal Society of Chemistry Where he talks of Lux numbers here that is the amount of light that has transmitted through the coloured liquid. The higher the lux number the lower the Absorbance value - which is the value we normally refer to for Advanced Higher …
Some experiments to try using Colorimetry:
- Determining Manganese Content in Fertiliser using Colorimetry https://www.canterbury.ac.nz/media/documents/science-outreach/manganese_fertiliser.pdf
- Iron Colorimetry Experiment: https://www.canterbury.ac.nz/media/documents/science-outreach/iron_colorimeter.pdf
- Measuring Calcium and Magnesium content of things like sea water or milk: https://www.canterbury.ac.nz/media/documents/science-outreach/magnesium_calcium.pdf
Calcium content in milk - complexometric titration and colorimetry: https://www.canterbury.ac.nz/media/documents/science-outreach/calcium.pdf
Beer Lambert Law / Absorbance - good explanation - though may be a little more information than you need for Adv H Chemistry ....Go to Reference
A bit about Colour !
In chemistry and pigments, the following happens ...
- A Black object absorbs all the colors of the visible spectrum and reflects none of them to the eyes. (think of a black object or clothes on a hot summer day - they absorb everything and get hot)
- White reflects all the colours of the visible light spectrum to the eyes (their atomic bonds do not absorb any frequencies). (white clothes are ideal for a summer's day as they reflect all the heat)
- Colourless objects do not reflect the light, they are transparent to light, letting all frequencies pass
If you want to read more about colour - and pigments - dyes and art etc then there is loads to read here - but its mostly just for interest - mostly not in the Adv H Chem course -
http://www.rsc.org/learn-chemistry/resources/art/colour#/ff3300
Bonding - some resonance structures : (Not sure if these are definitely in the course but probably worth looking at, even if just for some general knowledge)
Lewis dot diagrams :http://www.rsc.org/learn-chemistry/resources/art/colour#/ff3300
Bonding - some resonance structures : (Not sure if these are definitely in the course but probably worth looking at, even if just for some general knowledge)
http://www.mikeblaber.org/oldwine/chm1045/notes/Bonding/Resonan/Bond07.htm
Interesting film about Forensic Science
Physics solving Crimes ! Using an Ion beam of X-rays and Gamma rays to obtain a spectrum of gunshot residue :
http://www.youtube.com/watch?v=JgbdaQWfjDk
Transition Metal compounds
Colourful manganese and vanadium compounds - colour varies depending on oxidation state of the compounds they are involved in:
Colour changing in Manganese
Vanadium colour changes
The Colours of Complex Metal Ions
This is worth printing out to keep and read:
http://www.chemguide.co.uk/inorganic/complexions/colour.html
Another similar reference - worth a read through - about colours of complexes etc.
http://chem.libretexts.org/Core/Inorganic_Chemistry/Coordination_Chemistry/Complex_Ion_Chemistry/Origin_of_Color_in_Complex_Ions
Colour Chart
Here is a nice chart of the colours of the different transition metals, depending on oxidation states - you don't need to know all these colours but its something colourful to hang on your wall and remind you that they do change colour depending on what ligands surround them http://www.compoundchem.com/wp-content/uploads/2014/03/Transition-Metal-Ion-Colours-Aqueous-Complexes.pngAnother similar reference - worth a read through - about colours of complexes etc.
http://chem.libretexts.org/Core/Inorganic_Chemistry/Coordination_Chemistry/Complex_Ion_Chemistry/Origin_of_Color_in_Complex_Ions
Colour Chart
Coloured Glass - another nice picture to print out : http://www.compoundchem.com/wp-content/uploads/2015/03/The-Chemistry-of-Coloured-Glass.png
- a video about colouring glass - https://www.youtube.com/watch?v=FJ4PGwDtSMs
- Here are some rules and some questions about naming complex ions that are made up of a central metal ion and ligands http://www.milngavietutors.com/p/naming-complex-ions-and-questions.html
- Rules summarised on a graphic
- I have a good reference document if you need more information get in touch
- Complicated reference if you want to read more but way beyond Adv H level https://iupac.org/wp-content/uploads/2018/05/Inorganic-Brief-Guide-V1-3.pdf
EDTA - a hexadentate ligand with all sorts of uses - an interesting page from the Royal Society of Chemistry's excellent online magazine The Mole (PRINT THIS OUT TO KEEP !)
http://www.rsc.org/images/TM0313%20Magnificent%20Molecules%20-%20EDTA_tcm18-230873.pdf
Ligands etc - bidentate, hexadentate - useful information - http://www.chemguide.co.uk/inorganic/complexions/whatis.html
Schweizer's Reagent - This is just a snippet of interest - one of the ligand complexes you might have named - and its use in making cellulose etc https://en.wikipedia.org/wiki/Schweizer%27s_reagent
Drawing Lewis Structures - electrons to create molecular structure (not sure if this is in CfE course...)
http://www.chem.ucla.edu/harding/lewisdots.html
Splitting of D orbitals - nice example at the bottom here :
https://www.wou.edu/las/physci/ch462/tmcolors.htm
Here is a very good resource about ligands, colour and splitting of D Orbitals :
http://alevelchem.com/aqa_a_level_chemistry/unit3.5/s354/04.htm
Quiz about Ligands at the bottom of this page -
http://www.a-levelchemistry.co.uk/AQA%20A2%20Chemistry/Unit%205/5.4%20Transition%20Metals/5.4%20Transition%20metals%20home.htm
Try 5.4 part 1 and can check answers
Do not try 5.4 part 2 at this stage as it is not all relevant
UV/Vis Spectroscopy Video
http://www.youtube.com/watch?v=O39avevqndU
Catalysis
Fabulous video explaining the Cobalt 2+ to Cobalt 3+ being used as a catalyst as featured in Bright Red book page 25 - well explained video - watch it even if you already think you know about it !
https://www.youtube.com/watch?v=5neq2HVajoA
(Extra bits - not in the CfE course now from 2014 onwards
Ionic Crystal Lattice Structures (not in Revised for 2014....)
You can rotate and examine the CsCl (8:8 ratio) and NaCl (6:6 ratio) typical crystalline structures here :http://www.neubert.net/Crystals/CRYStruc.html
Chlorides (not in Revised....)
http://www.chemguide.co.uk/inorganic/period3/chlorides.html )
http://www.rsc.org/images/TM0313%20Magnificent%20Molecules%20-%20EDTA_tcm18-230873.pdf
Ligands etc - bidentate, hexadentate - useful information - http://www.chemguide.co.uk/inorganic/complexions/whatis.html
Schweizer's Reagent - This is just a snippet of interest - one of the ligand complexes you might have named - and its use in making cellulose etc https://en.wikipedia.org/wiki/Schweizer%27s_reagent
Drawing Lewis Structures - electrons to create molecular structure (not sure if this is in CfE course...)
http://www.chem.ucla.edu/harding/lewisdots.html
Splitting of D orbitals - nice example at the bottom here :
https://www.wou.edu/las/physci/ch462/tmcolors.htm
Here is a very good resource about ligands, colour and splitting of D Orbitals :
http://alevelchem.com/aqa_a_level_chemistry/unit3.5/s354/04.htm
Quiz about Ligands at the bottom of this page -
http://www.a-levelchemistry.co.uk/AQA%20A2%20Chemistry/Unit%205/5.4%20Transition%20Metals/5.4%20Transition%20metals%20home.htm
Try 5.4 part 1 and can check answers
Do not try 5.4 part 2 at this stage as it is not all relevant
UV/Vis Spectroscopy Video
http://www.youtube.com/watch?v=O39avevqndU
Catalysis
Fabulous video explaining the Cobalt 2+ to Cobalt 3+ being used as a catalyst as featured in Bright Red book page 25 - well explained video - watch it even if you already think you know about it !
https://www.youtube.com/watch?v=5neq2HVajoA
(Extra bits - not in the CfE course now from 2014 onwards
Ionic Crystal Lattice Structures (not in Revised for 2014....)
You can rotate and examine the CsCl (8:8 ratio) and NaCl (6:6 ratio) typical crystalline structures here :http://www.neubert.net/Crystals/CRYStruc.html
Chlorides (not in Revised....)
http://www.chemguide.co.uk/inorganic/period3/chlorides.html )





